INDICATION
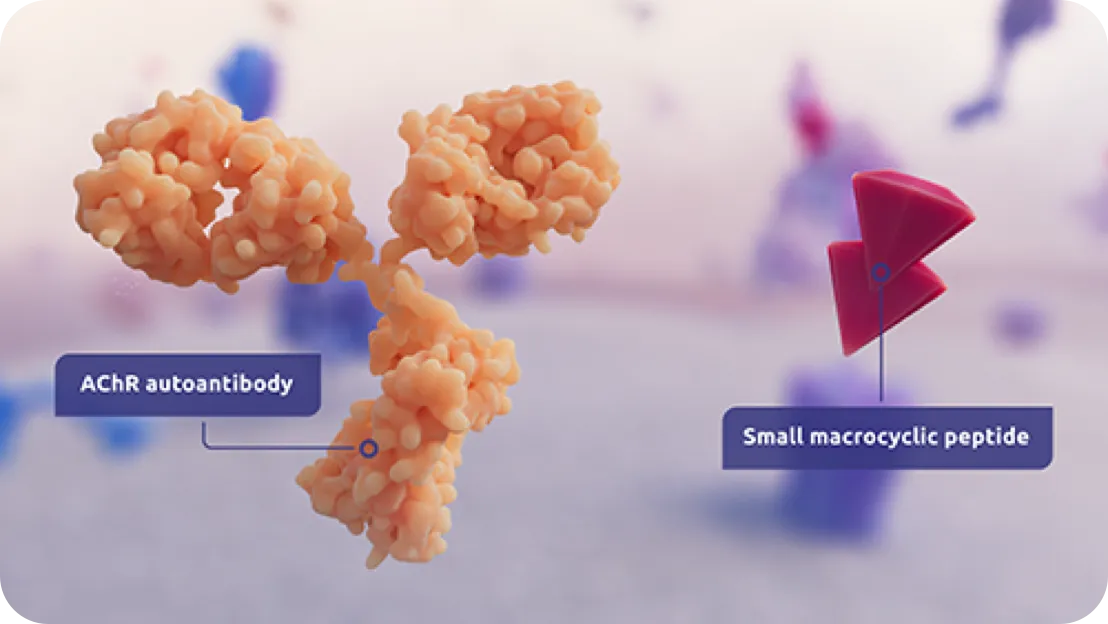
ZILBRYSQ (zilucoplan) is indicated for the treatment of generalized myasthenia gravis (gMG) in adult patients who are anti-acetylcholine receptor (AChR) antibody positive.
IMPORTANT SAFETY INFORMATION INCLUDING BOXED WARNING
WARNING: SERIOUS MENINGOCOCCAL INFECTIONS
ZILBRYSQ, a complement inhibitor, increases the risk of serious infections caused by Neisseria meningitidis. Life-threatening and fatal meningococcal infections have occurred in patients treated with complement inhibitors. These infections may become rapidly life-threatening or fatal if not recognized and treated early.
-
Complete or update vaccination for meningococcal bacteria (for serogroups A, C, W, Y, and B) at least 2 weeks prior to the first dose of ZILBRYSQ, unless the risks of delaying therapy outweigh the risk of developing a serious infection. Comply with the most current Advisory Committee on Immunization Practices (ACIP) recommendations for vaccination against meningococcal bacteria in patients receiving a complement inhibitor.
-
Patients receiving ZILBRYSQ are at increased risk for invasive disease caused by Neisseria meningitidis, even if they develop antibodies following vaccination. Monitor patients for early signs and symptoms of serious meningococcal infections and evaluate immediately if infection is suspected.
Because of the risk of serious meningococcal infections, ZILBRYSQ is available only through a restricted program under a Risk Evaluation and Mitigation Strategy (REMS) called ZILBRYSQ REMS.
CONTRAINDICATIONS
ZILBRYSQ is contraindicated for initiation in patients with unresolved serious Neisseria meningitidis infection.
WARNINGS AND PRECAUTIONS
Serious Meningococcal Infections
ZILBRYSQ, a complement inhibitor, increases a patient’s susceptibility to serious, life-threatening, or fatal infections caused by meningococcal bacteria (septicemia and/or meningitis) in any serogroup, including non-groupable strains. Life-threatening and fatal meningococcal infections have occurred in both vaccinated and unvaccinated patients treated with complement inhibitors. The initiation of ZILBRYSQ treatment is contraindicated in patients with unresolved serious Neisseria meningitidis infection.
Complete or update meningococcal vaccination (for serogroups A, C, W, Y, and B) at least 2 weeks prior to administration of the first dose of ZILBRYSQ, according to current ACIP recommendations for patients receiving a complement inhibitor.
If urgent ZILBRYSQ therapy is indicated in a patient who is not up to date with meningococcal vaccines according to ACIP recommendations, provide the patient with antibacterial drug prophylaxis and administer meningococcal vaccines as soon as possible.
Closely monitor patients for early signs and symptoms of meningococcal infection and evaluate patients immediately if infection is suspected. Consider interruption of ZILBRYSQ in patients who are undergoing treatment for serious meningococcal infection, depending on the risks of interrupting treatment in the disease being treated.
ZILBRYSQ REMS
Due to the risk of serious meningococcal infections, ZILBRYSQ is available only through a restricted program under a REMS called ZILBRYSQ REMS.
Under the ZILBRYSQ REMS, prescribers must enroll in the program. Prescribers must counsel patients about the risk of serious meningococcal infection, provide the patients with the REMS educational materials, and ensure patients are vaccinated with meningococcal vaccines. Additional information on the REMS requirements is available at www.ZILBRYSQREMS.com or 1-877-414-8353.
Other Infections
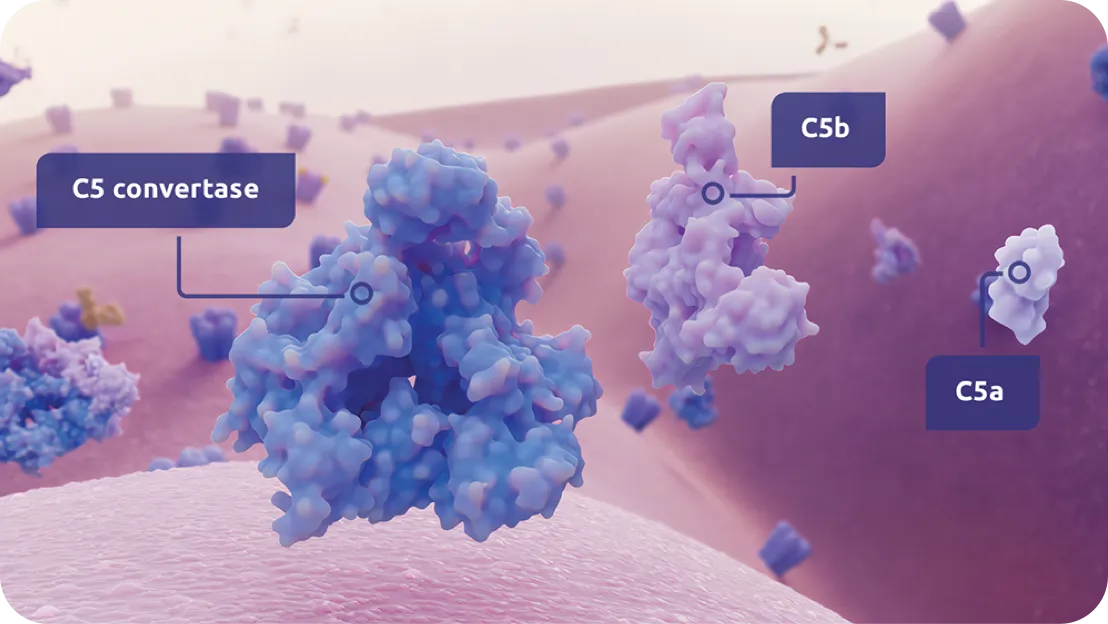
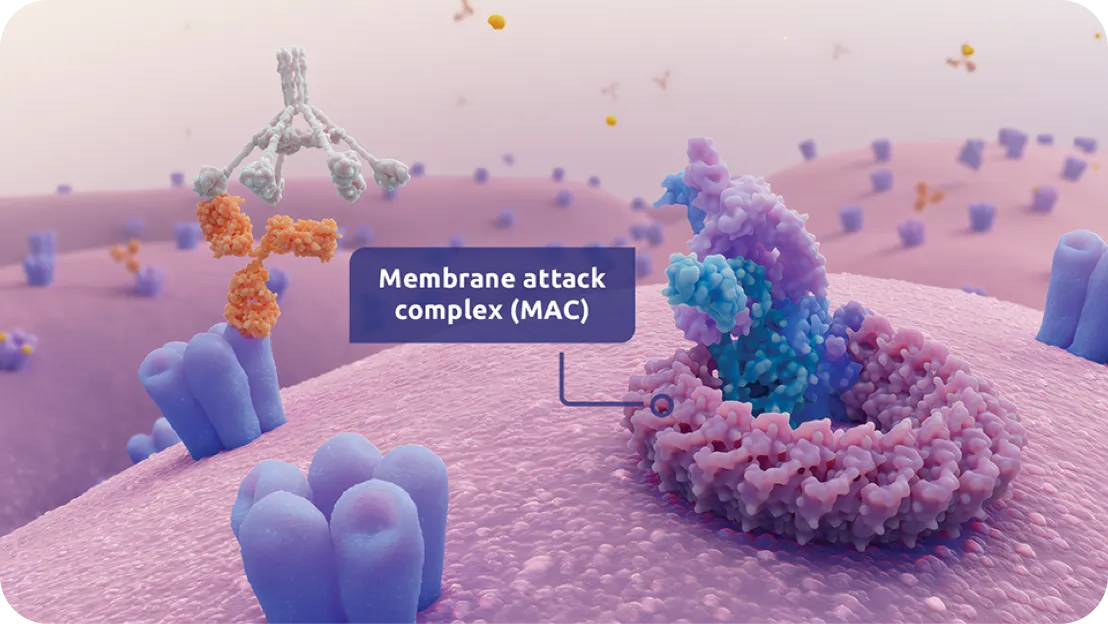
Serious infections with Neisseria species (other than Neisseria meningitidis), including disseminated gonococcal infections, have been reported in patients treated with complement inhibitors. ZILBRYSQ blocks terminal complement activation; therefore, patients may have increased susceptibility to infections, especially with encapsulated bacteria, such as infections caused by Neisseria meningitidis but also Streptococcus pneumoniae, Haemophilus influenzae, and to a lesser extent, Neisseria gonorrhoeae. Administer vaccinations for the prevention of Streptococcus pneumoniae infection according to ACIP recommendations. Patients receiving ZILBRYSQ are at increased risk for infections due to these organisms, even if they develop antibodies following vaccination.
Pancreatitis and Other Pancreatic Conditions
Pancreatitis and pancreatic cysts have been reported in patients treated with ZILBRYSQ. Patients should be informed of this risk before starting ZILBRYSQ. Obtain lipase and amylase levels at baseline before starting treatment with ZILBRYSQ. Discontinue ZILBRYSQ in patients with suspected pancreatitis and initiate appropriate management until pancreatitis is ruled out or has resolved.
ADVERSE REACTIONS
In a placebo-controlled study, the most common adverse reactions (reported in at least 10% of gMG patients treated with ZILBRYSQ) were injection site reactions, upper respiratory tract infections, and diarrhea.
Please see the full Prescribing Information for additional Important Safety Information.